Basic of Steam : Characteristic of Steam
Steam is tasteless, odorless, colorless, and invisible gas which is generated when water is heated by boiler.
The generating process of steam is shown in the figures below.
To raise temperature of water to the boiling point, it is necessary to add sufficient thermal energy.
Furthermore, to change water to steam, without raising temperature more, it is necessary to add more thermal energy.
The energy raises water to the boiling point is called as water quantity of heat or sensible heat, and the energy changes boiling water to steam is called as latent heat.
This heat quantity is changed with conditions of pressure and temperature.
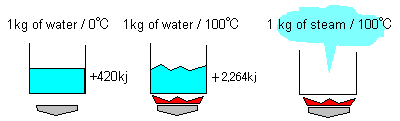
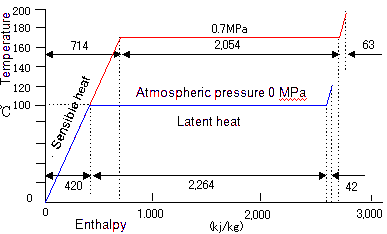
We show heat quantity required to generate steam from 1 kg of water under atmospheric pressure.
The sensible heat of 4.2 kj is needed to raise temperature by 1 ℃ until it reaches the boiling point.
To make water at 100 ℃ to steam at 100 ℃, the latent heat of 2,264kj is needed.
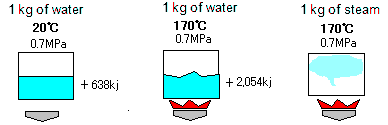
We show heat quantity to generate 1 kg of steam under pressure of 0.7 MPa.
Compared with under atmospheric pressure, the boiling point is higher, the sensible heat to boil water is higher, and the latent heat for changing water to steam is smaller.
All the heat quantity (= sensible heat + latent heat) for generating 1 kg of steam is needed more than that in atmospheric pressure.